|
Individually, each protein primarily plays a role in the structure of the virus particle, but they are also involved in other aspects of the replication cycle. 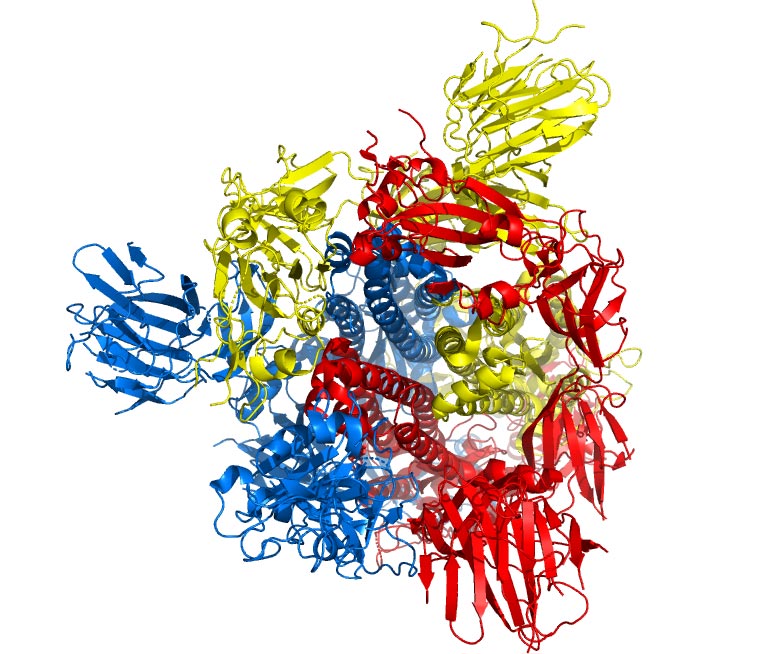
More recently, however, it has become clear that some CoVs do not require the full ensemble of structural proteins to form a complete, infectious virion, suggesting that some structural proteins might be dispensable or that these CoVs might encode additional proteins with overlapping compensatory functions. The coronaviral genome encodes four major structural proteins: the spike (S) protein, nucleocapsid (N) protein, membrane (M) protein, and the envelope (E) protein, all of which are required to produce a structurally complete viral particle. Live, attenuated vaccines and fusion inhibitors have proven promising, but both also require an intimate knowledge of CoV molecular biology. Clearly, the seriousness of these infections and the lack of effective, licensed treatments for CoV infections underpin the need for a more detailed and comprehensive understanding of coronaviral molecular biology, with a specific focus on both their structural proteins as well as their accessory proteins. Since the outbreak of MERS in April 2012 up until October 2018, 2229 laboratory-confirmed cases have been reported globally, including 791 associated deaths with a case-fatality rate of 35.5%. The consequent outbreak of SARS in 2003 led to a near pandemic with 8096 cases and 774 deaths reported worldwide, resulting in a fatality rate of 9.6%. Accordingly, these cross-species barrier jumps allowed CoVs like the SARS-CoV and Middle Eastern respiratory syndrome (MERS)-CoV to manifest as virulent human viruses. Some CoVs were originally found as enzootic infections, limited only to their natural animal hosts, but have crossed the animal-human species barrier and progressed to establish zoonotic diseases in humans. In recent years, it has become increasingly evident that human CoVs (HCoVs) are implicated in both URTIs and LRTIs, validating the importance of coronaviral research as agents of severe respiratory illnesses. They can also infect humans and cause disease to varying degrees, from upper respiratory tract infections (URTIs) resembling the common cold, to lower respiratory tract infections (LRTIs) such as bronchitis, pneumonia, and even severe acute respiratory syndrome (SARS). Coronaviruses primarily infect birds and mammals, causing a variety of lethal diseases that particularly impact the farming industry. Based on genetic and antigenic criteria, CoVs have been organised into three groups: α-CoVs, β-CoVs, and γ-CoVs (Table 1). With genome sizes ranging from 26 to 32 kilobases (kb) in length, CoVs have the largest genomes for RNA viruses. The high mortality rate of certain CoVs, along with their ease of transmission, underpins the need for more research into CoV molecular biology which can aid in the production of effective anti-coronaviral agents for both human CoVs and enzootic CoVs.Ĭoronaviruses (CoVs) (order Nidovirales, family Coronaviridae, subfamily Coronavirinae) are enveloped viruses with a positive sense, single-stranded RNA genome. Data shows that E is involved in critical aspects of the viral life cycle and that CoVs lacking E make promising vaccine candidates. 
The most progress has been made on SARS-CoV E, highlighting specific structural requirements for its functions in the CoV life cycle as well as mechanisms behind its pathogenesis. Good progress has been made but much still remains unknown and this review has identified some gaps in the current knowledge and made suggestions for consideration in future research. It also compares E to other viral proteins of a similar nature to speculate the relevance of these new findings. This review aims to establish the current knowledge on CoV E by highlighting the recent progress that has been made and comparing it to previous knowledge. Recent studies have expanded on its structural motifs and topology, its functions as an ion-channelling viroporin, and its interactions with both other CoV proteins and host cell proteins. The CoV envelope (E) protein is a small, integral membrane protein involved in several aspects of the virus’ life cycle, such as assembly, budding, envelope formation, and pathogenesis. A renewed interest in coronaviral research has led to the discovery of several novel human CoVs and since then much progress has been made in understanding the CoV life cycle. The outbreak of severe acute respiratory syndrome (SARS) in 2003 and, more recently, Middle-East respiratory syndrome (MERS) has demonstrated the lethality of CoVs when they cross the species barrier and infect humans. Coronaviruses (CoVs) primarily cause enzootic infections in birds and mammals but, in the last few decades, have shown to be capable of infecting humans as well.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
